
MEDRAD® Intego
MEDRAD® Intego
Safety features, accuracy and efficiency. All in one system.
MEDRAD® Intego allows accurate and personalized dose-on-demand administration of 18F-FDG and 18F-NaF from a multi-dose vial, while shielding and protecting the operator.
How does MEDRAD® Intego work?
A multi-dose vial of 18F-FDG or 18F-NaF is delivered to the nuclear medicine department. The operator loads the custom tungsten vial shield into the system and installs the source administration set (SAS).
After the system is primed and the concentration of radiopharmaceutical is checked, the operator can set the next patient injection protocol (weight-based or fixed dose) and inject the radiopharmaceutical. The system also allows for an optional saline test injection
The patient administration set (PAS) must be changed with each patient. The SAS must be changed with new radiopharmaceutical vial.
Demo & Videos
Watch the following videos for a user-friendly demonstration of the MEDRAD® Intego system and basic set-up.
What Are the Benefits of MEDRAD® Intego?
Safety Features for Clinicians: Reduction in Radiation Exposure
- Automated dose preparation and infusion enable hands-off operation and increased distance from the activity source
- Tungsten and lead shielding reduce radiation exposure for clinicians
- Clinical experience shows a reduction in radiation exposure compared to manual infusion. For example, studies have found*:
*Please refer to the Clinical reference brochure for more published experiences.
Personalized Patient Care
- Automated weight-based dosing from a easy to use touchscreen enables clinicians to administer an accurate, personalized dose for each patient
- Variable flow rate (0.5 mL/sec or 1 mL/sec). The lower flow rate may be especially useful in patients with fragile veins
- Saline test injection (optional) to verify the patency of the IV device
Operational Efficiency
The MEDRAD® Intego PET Infusion system can help streamline your workflow and drive operational efficiency, thanks to:
- Elimination of non-value-added steps with automated dose preparation, administration, and documentation
- Schedule flexibility to respond to patients who arrive late or require unexpected prep time with dose-on-demand functionality from a multi-dose vial
- MEDRAD® Intego Connect (optional):
- Enables wireless schedule import for streamlined schedule management
- Offers automated wireless export of infusion records and radiation dose to PACS*
- Supports quality initiatives with reliable and comprehensive information from point of care to study interpretation
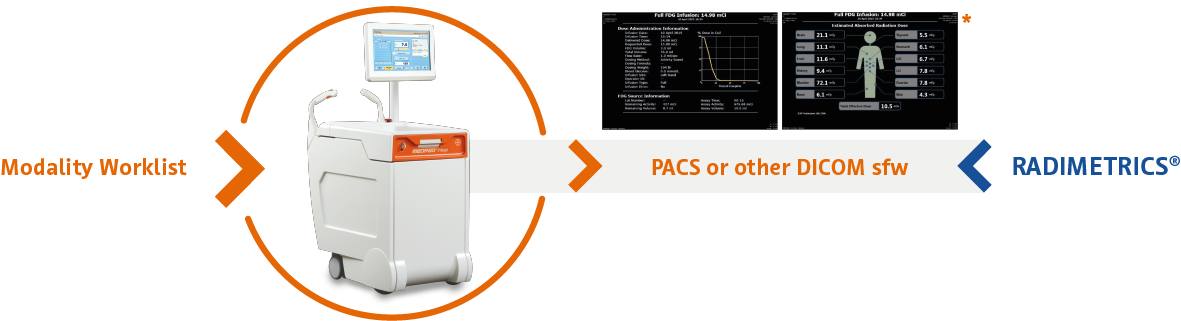
* Absorbed radiation dose is calculated using ICRP 106 (from software version 18) for 18F-FDG and NUREG/CR-6345 for 18F-NaF. In compliance with HIPAA regulations, patient cases included in the GUIs are fictitious examples only and do not contain any actual patient data. Import and export functionalities and RADIMETRICS® are sold as a separate option. Output could be sent also to other destination configured for reading DICOM
One System, 6 Million Proven Results
MEDRAD® Intego has been trusted for PET infusions more than 6 million times around the world.
Worldwide cumulative infusions with MEDRAD® Intego
Information
| Weight | Approximately 355 kg (783 lbs) |
| Dimensions | Approximately 50.8 cm (20") wide, 82.5 cm (32.5") deep, 127.5 cm (50") high with POC-615 display |
| Maximum radioactivity | 27.7 GBq (750 mCi) for shielding effectiveness 25.9 GBq (700 mCi) for dose preparation |
| Maximum RP volume | 3700 MBq/mL (100 mCi/mL) |
| Dose range | 37-925 MBq (1-25 mCi) |
| +/- 2% of the measured dose, exclusive of dose calibrator calibration factor | |
| Flow rate | 0.5 mL/sec or 1 mL/sec |
| System radiation profile | Meets the NRC Code of Federal Regulations for Public Exposure, which limits radiation exposure to 0.02mSv (2mrem) in any one hour period. With the maximum activity loaded, the maximum radiation exposure at the front, rear, left, right, and top of the cart does not exceed 0.02mSv/hr (2mrem/hr) when the system is idle. The maximum radiation exposure at the front, rear, left, right, and top of the cart does not exceed 0.05mSv/hr (5mrem/hr) while holding a prepared dose prior to delivery to the patient. |
| Power consumption, maximum | 300 VA |
| Voltage | 100-240 VAC |
| Frequency | 50/60 Hz |
| Product | Catalog |
|---|---|
| MEDRAD® Intego PET Infusion System | INT SYS 200 |
| MEDRAD® Intego Connect includes the software licenses catalog numbers | SW - WLIST, SW - IRPACS and SW - RDPACS |
| Source Administration Set | (SAS) INT CSS |
| Patient Administration Set | (PAS) INT CPS |
| Replacement Printer Label Roll | 60937964 |
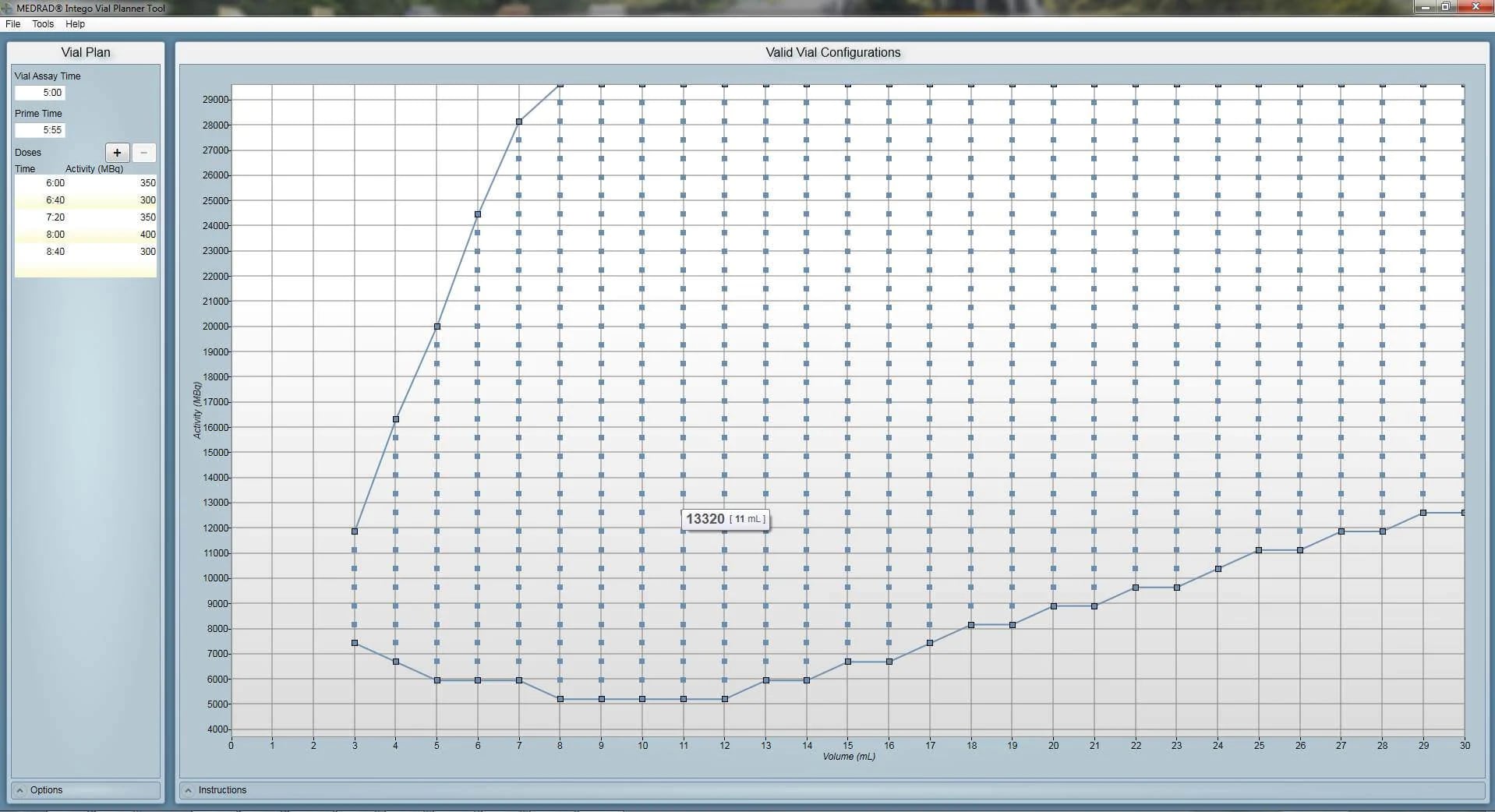
Vial Planner Tool
This tool is intended to calculate the approximate amount of 18F-FDG or 18F-NaF needed to complete a patient schedule with the MEDRAD® Intego PET Infusion System. The tool also allows observation of the potential impact of variations to the schedule plan.
Note: The displayed information is fictitious and does not represent any specific patient or dosing data.
Downloads
References
1. Lecchi M, Lucignani G, Maioli C, Ignelzi G, Del Sole A, “Validation of a new protocol for ¹⁸F-FDG infusion using an automatic combined dispenser and injector system,” Eur J Nucl Med Mol Imaging 39, no. 11 (2012): 1720-1729.
2. Sánchez RM, Vano E, Fernández JM, Ginjaume M, Carreras JL, “Evaluation of an automated FDG dose infuser to PET-CT patients,” Radiat Prot Dosimetry 165, no. 1-4 (2015): 457-460.
3. Based on Worldwide Patient Administration Set sales data August 2008 – May 2022.
4. Based on Worldwide Patient Administration Set sales in 2021 and 250 working days.






